My SciELO
Estudos Sociedade e Agricultura
Print version ISSN 1413-0580
Estud.soc.agric. vol.5 no.se Rio de Janeiro 2010
Labeling as precaution: the release of RR soy and the regulation of transgenics in Brazil
Cristiano Luis Lenzi1
Translated by Celina de Castro Frade
Translation from Estudos Sociedade e Agricultura, Rio de Janeiro, vol. 18 no. 1, p. 220-255, Abril 2010.
ABSTRACT
This paper presents an analysis of the conflict involving the release of Roundup Ready (RR) soy in Brazil from what is called a story-line of labeling. The aim is to assess the resources and strategies used by two discursive alliances existing in the conflict to legitimize their position in the political arena in the process of release of transgenics. The work examined statements and arguments that permeated the debate on the issue in the country at different times and used the case involving the release of RR soy as a reference for the analysis. The text aims to show that these discourses have very different assumptions about labeling and its relation to issues involving the principle of substantial equivalence, science, risk and nutritional safety.
Key words: labeling- GMOs- precaution- discourse- conflict.
"When I presented this project [labeling], I did not intend to define all the policy about transgenics in Brazil. ( ). Obviously, it was not only a matter of consumer's choice, it was based on the principle of precaution ( ). (Representative Fernando Gabeira). "
"The government has not decided for labeling because it thinks it is dangerous. The government has decided for labeling because it thinks it is the consumer's right to know if the product he is consuming is a transgenic product (Bresser Pereira, former Science and Technology - S&T - Minister).
For the last six or seven years, by regulating transgenics, the governors and even the scientific community have, all the time, committed themselves to the idea that the consumer would be ensured his/her right to information so as he/she could then perform his/her legitimate right to eat or not to eat. All these promises, as we know, have not been fulfilled. The real truth is that, in the end, there has never been so far any labeling for transgenic products in Brazil" (Luiz Eduardo R. de Carvalho - former president of the Brazilian Society of Food Science and Technology).
Introduction
In the last decade, a growing number of countries started to implement policies for transgenics labeling. Moreover, in addition to labeling itself, conflicts and tensions usually resulted from this kind of policy became very common in these countries. For the companies, labeling is a key element in the product's marketing policy and tends to be seen as directly influencing consumer's decisions. On the other hand, for environmentalists, labeling tends to be seen as a space of symbolic struggle and a means of reaching a better regulation of these products. Due to this, it seems natural that labeling constitutes itself an important space for the struggles that seek to define the commerce of genetically modified organisms (GMO).
In this paper we will make an attempt to discuss the development of this conflict in Brazil in different moments with reference to RR soy. The conflict is discussed from what we call a storyline of labeling. This represents the axe through which the various structuring issues of conflict over labeling present themselves in the Brazilian case. To discuss these issues, we will make use of an interpretive analysis of the political process. Under this approach, politics is seen as a continuous process of discursive struggle for the definition of social and political problems.
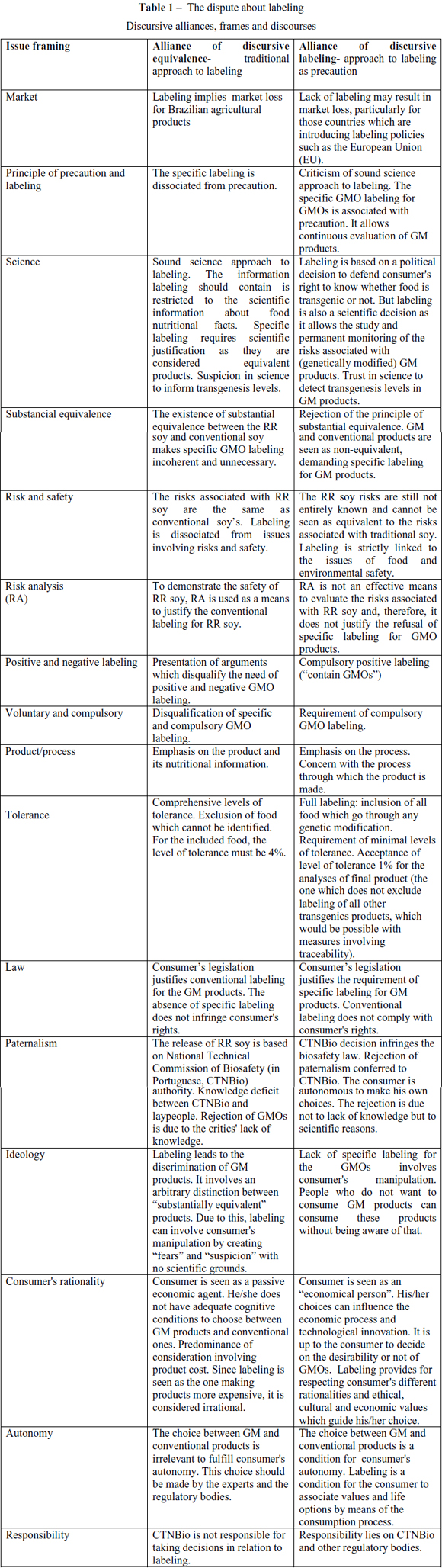
The key issue for the interpretive political analysis is how the political issue is being conceptualized or framed in the political debate (FISCHER, 2003). Hajer's (1995) concept of story-line is a way of examining the frames embedded in such conflicts. In The Politics of Environmental Discourses, the author shows that storylines "are devices through which actors are positioned, and through which specific ideas of "blame" and "responsibility", and of "urgency" and "responsible behaviour" are attributed" (HAJER, 1995:65). Under an environmental controversy, says the author, the discursive elements are presented as "narratives on social reality through which elements from many different domains are combined and that provide actors with a set of symbolic references that suggest a common understanding" (HAJER, 1995: 62). The labeling storyline is formed by two discursive alliances: a) the discursive alliance of labeling and b) the discursive analysis of substantial equivalence. Such discourses display different assumptions on labeling and its relation with issues involving the principle of substantial equivalence, labeling, science, risk and consumer's choice (see Table 1). The other part of the study aims at examining these differences.
Labeling risks: the release of commercial RR soy and the conflict over labeling
Differently from the discussion about the use of Risk Analysis (RA) in the release of RR soy, the debate on labeling seemed to offer a more consensual picture for the GMO critics and supporters in Brazil. After all, since the beginning, representatives of CTNBio and the government seemed favorable, at least in discourse, to labeling. But if there is an apparent consensus for labeling GM products, why did labeling raise such an intense conflict between the government and civil society groups?
Firstly, it is worth considering that since the beginning of CTNBio, organizations such as Institute of Consumer Protection (in Portuguese,IDEC) and Greenpeace have not seen any serious commitment of the biosafety commissions to labeling policy. In 1996, IDEC sent various requests to the commission, including the creation of a labeling policy, without any answer. That is why this organization separated from the biosafety commission in 1997. This lack of interest was based on the own limits of responsibility that the CTNBio members assigned to the commission in its role to implement a labeling policy. For Esper Cavalheiro, former president of CTNBio: "The labeling issue transcends CTNBio legal responsibilities as it involves issues related to consumer's protection and so it is related to the Consumer Protection Code". For Leila Oda, labeling is "more a political issue than a technical one". So, as far as CTNBio was seen as a scientific commission, labeling itself, as a "political" project, was seen as something strange to the commission, which shows that the issues involved the labeling of GMO products were not regarded as CTNBio own responsibility.
Moreover, the release of soy growing, as it seems, would lead to a gradual and automatic commercial release of the product. The approval occurred, on the other hand, without the government presenting any specific labeling program for the GMOs, resulting in an immediate reaction from civil society organizations. In the public civil lawsuit brought against the government and CTNBio, IDEC claimed then that before analyzing and issuing a conclusive technical opinion to Monsanto's request, the government should regulate the "norms of food safety, trade and labeling. Without it, it cannot evaluate any request" [our emphasis] (IDEC, 2008). We could assume that the release for the growing would wait for further studies to confirm the safety of soy. However, this assumption was constantly denied by CTNBio and the government, which started to see risk analysis (RA) as a satisfactory means to evaluate the risks linked to RR soy. If the safety of soy had already been confirmed by RA, what would be the obstruction to release it commercially afterwards?
The import of transgenic soy proved itself another important issue for the development of the conflict. This launched a shadow of doubts on the real intentions of the government or on its capacity to conduct the regulation of transgenics in the country. One should remember that both S&T Ministry and CTNBio did not take any measures to reverse what had happened and even tried to inform the resistance to the fact in regard the regulatory policy which was being implemented.
Labeling safety: defending the consumer's right in the absence of danger
In the conflict about transgenic labeling, the alliance of discursive equivalence shows a position that can be found in other countries. In the USA, for example, the Biotechnology Industry Organization (BIO) has informed that it is in favor of the consumer's right though, at the same time, was against compulsory labeling because it consider it could confuse consumers (KLINTMAN, 2002). In Brazil, it was not uncommon that members of the government, CTNBio and the food industry become favorable to the consumer's right, but it was not uncommon either to present arguments to invalidate the implementation of specific labeling for the GM products.2 To analyze this ambivalence, we need to examine the different arguments generally shown to invalidate transgenic labeling.
Different reasons are usually claimed for not labeling transgenics. The economic costs associated with labeling are usually presented as a way to reject a specific labeling system for these products. Once labeling can raise the GMOs' prices, the economic benefits associated with them could be made void for the consumer. This could happen, particularly if the costs were enough to enable the offer of such products in the market. Another argument is based on the thesis that labeling would also involve an arbitrary distinction among "substantially equivalent" products. This argument unfolds into a general approach which claims that, once GM and conventional products display equivalent nutritional proprieties, there would be no legal grounds for justifying their labeling. Therefore, if there is no scientific or legal grounds to differentiate GM and conventional products, labeling could create "guilt by association". Products would be considered more dangerous though presenting themselves, in terms of safety, similar to the others.
It is also claimed that labeling tends to be confused, erroneous and irrelevant even when its information is considered correct. An American scientist contrary to labeling says that, for example, "even a message that is accurate, in the narrowest sense, can mislead and confuse consumers if it is irrelevant, unintelligible, or so craftily selected that it provides inadequate or biased information" (MILLER apud KLINTMAN, 2002:74). Finally, the supporters of non-labeling also based on the impossibility of evaluating the levels of transgenesis showing the technical and scientific impossibilities to reach this goal.
In the Brazilian conflict, there were not few moments in which the arguments for discrediting labeling were presented. In spite of, in discourse, they were for consumer's right, it was not uncommon representatives of the government, CTNBio and the food industry help to promote some of these arguments which are just used to discredit a specific labeling system for the GM products. The thesis of Bresser-Pereira and CTNBio that soy was a "substantially equivalent" product, for example, is the basis of the Federal Drug Administration (FDA) discourse in the United States of America (USA) for exactly not labeling these products.3 In 1999, after justice enforced the need to conduct a specific labeling policy for the GM products, various arguments contrary to labeling were raised again in public hearings. It was expressed, for example, by representative Luciano Pizzato who showed that, in the absence of scientific facts about the risks of transgenic soy, labeling would be incoherent (PIZATO, 2000: 17). So, although the rejection of labeling has not always been evident in the discursive alliance of equivalence, the criticism to its irrationality always proves itself in a present aspect in this discourse.
Their proponents accuse the informative inconsistency provided by labeling and its little validity to promote consumer's rational choice. In the former, it will be based on the classificatory arbitrariness that labeling establishes among "substantially equivalent" products. For representative Confúcio Moura, the labeling bill introduced by representative Gabeira in 1999, which foresaw the labeling of the GM products, looks controversial "because the countries, particularly the USA, find it discriminatory the placement of specific labeling". Labeling also would be inconsistent by the "difficulties of indicating the ingredients and the sub-products in food composition". Therefore, for the representative, "labeling would be a very difficult and discriminatory way for certain products" (MOURA, 2001: 12).
It is possible to notice in these extracts how the principle of substantial equivalence was used to invalidate the need for labeling. When the former president of CTNBio, Dr. Barreto de Castro, says that, not being identified the risks of soy, it would be irrelevant the requirement for labeling, he is also using in his discourse the principle of substantial equivalence since the risks of soy were examined out of the assumptions of such principle. And so does Bresser-Pereira, former S&T Minister, by saying that, in the case of soy, "there is no substantial change of the product" and that the "product is exactly the same"4 This principle plays a key role in the conflict about labeling in Brazil and evidences that the assumptions used by CTNBio to guide its decision for the commercial release resulted in direct implications for the debate on labeling in the country.
It is this principle that, somehow also sustains the accusations of discrimination. Because discrimination guides itself on the accusation that labeling would be making equivalent things become different. This accusation will happen in both ways. Firstly, the discrimination is seen as existing in the comparison that can be made with other transgenic products, but not agricultural ones.5 The second type of discrimination is seen as associated with the type of communication provided for labeling itself. The refusal of the food industry to incorporate specific labeling for the GM products in the conflict was justified by the possible distorted communication that labeling would be about to produce. As informed by the Brazilian Association of Food Industry (in Portuguese, Abia) legal director, "[the labeling requirement] is in force, but it is not incorporated because industry does not want to associate its brand with an alert, as if it was something dangerous" (Abia Legal Director apud IDEC, 2008c). One concern that will also be present in the bill of the legislative decree n. 90, 2007, of representative Kátia Abreu, informing that the label to be placed in the products "refers to the idea of attention and care and can result in the population's suspicion about products which had already been evaluated and considered safe by the National Technical Commission of Biosafety (CTNBio), thus jeopardizing the insertion of these products in the market [our emphasis] (Abreu apud BRASIL, 2007).6
Some of these statements about the discrimination do not inform, however, where would be the communicative distortion they affirm to exist. Some arguments lead to thinking that, regardless how labeling presents itself, it will always lead unequivocally to consumer's irrationality. These criticisms do not always object to whether the message provided by labeling is true or false. They simply object to labeling by the simple irrational effect it can produce. Therefore, the "fear" and "suspicion" that it may create to consumers are seen as not having any valid scientific grounds for the consumer's decision-making.
Labeling as precaution: consumer's choice, autonomy and environmental safety
We can imagine many reasons through which people can get interested in labeling. The main reason takes us to the role of labeling in the process of consumer's choice. Labeling enables people to make choices as from questions associated with cultural prohibitions (vegetarianism and animal well-being) and health risks so as to articulate their consumption choices with their lifestyle. In this case, by offering a series of information associated with consumer's values, labeling can serve as a means to allow them to make autonomous choices. The discursive alliance of labeling has been based on arguments close to this one to justify the labeling of these products.
It is worth noticing that labeling was seen, at first, involving a kind of precaution. The liaison between labeling and precaution will be shown in this alliance in two distinct forms. One of them happens for a strategic reason when this discourse mentions the use of labeling as a means of postponing the release of soy and as a means of promoting protective effects with this action. In 2001, when Gabeira's labeling bill served as a basis for a initial discussion on labeling in the country, the representative informs that "it was not only a matter of consumer's choice, it was based on the principle of precaution". Thus, he found it "necessary to postpone a little the process of entrance of transgenics in Brazil" (GABEIRA, 2001: 02).
The decision of judge Antonio Souza Prudente in the civil lawsuit brought by IDEC and Greenpeace reveals a similar vision to the one precaution and labeling are also seen as interlinked. In this document, he points out that "The simple labeling of transgenic products is insufficient to fulfill the efficacy of the principle of prevention ( )" (PRUDENTE, 2000: 41). Although considered insufficient, labeling is seen here as an essential part of the principle. The judge mentions the insufficiency of labeling to apply the principle of prevention because it seeks exactly to establish a correlation between labeling and Environmental Impact Assessment (EIA) as a means of applying precaution. And following the same viewpoint, Lynn Silver, of the same organization, claims that "labeling is indispensable for the identification in the future of the adverse effects that may eventually arise after the introduction of the products". For her, "besides being the consumer's right, labeling is an essential tool for the control of unexpected effects as to human or even animal health" (SILVER, 2001: 25).
The relation between labeling and precaution is not always recognized as to the way the latter is usually expressed. Instead of absolute prohibition, precaution can be understood as enabling a wide range of measures that not always result in a prohibition of this kind. Precaution, as shown (WHITESIDE, 2006), may imply the need to implement permanent guard as, in many cases, it is not possible to reach an immediate decision about the safety of the products. Therefore, the more relevant contemporary measures in the field of transgenic labeling, imposing a compulsory labeling on all these products in the EU, have come out in a moment when it is recognized that the idea of precaution would require labeling and traceability of new products (WHITESIDE, 2006). The decision on opening space for corrective measures in the future is the core of the precaution policy and is this sense that, as seen before, is assigned to labeling by organizations such as IDEC.
The labeling alliance has also been supported by a legal argument and another one of moral character to approve the specific GMO labeling. The legal argument is based on the thesis that consumer's legislation would be in favor of labeling. Judge Souza Prudente's decision was basically based on this argument, In the civil lawsuit brought by IDEC, the judge affirms that:
"If it is undeniable that, in accordance with article 6, II and II, the consumer has the basic right to adequate and clear information, with correct specification of the characteristics, quality and risks presented, among other data, it is always right that only these data will provide for the adequate consumer's right of choice, also guaranteed by the Consumer Protection Code" (PRUDENTE, 2000).
Organizations such as Greenpeace and IDEC also saw in these rights a basic legal condition to justify the labeling of the GM products. However, this legal interpretation was put in doubt in another moment of the conflict. It was challenged by the decision of judge Selene Maria de Almeida on 25 February 2002 that issued an injunction that authorized the growing and commerce of Roundup Ready (RR) soy. The judge's decision, however, suspended judge Souza Prudente's decision and, this way, contradicted the interpretation which informs that the Consumer Protection Code would be enough to require the specific labeling of these products. This judge's decision is emblematic once it breaks with the usual interpretation that says that consumer's legislation would provide for the legal grounds to require the labeling of GM products.7
While the requirement of fulfilling EIAs was complied in the environmental legislation, the labeling requirement, in return, sought compliance in the consumer's legislation. This shows that, while the environmental legislation is clearer about the requirement of fulfilling EIAs for innovations that may result in some kind of impact for the environment, it is much less precise for the requirement of GMO labeling. In this process, the labeling requirement was at the mercy of the interpretation of the Consumer Protection Code and the way it can be interpreted to require the specific labeling for these products. While labeling supporters have seen in the consumer's legislation strong grounds to require labeling, representatives of the food industry have interpreted this legislation in their behalf. The reason for this contradiction seems to lie in the fact that, although consumer's legislation seems to provide a reason for labeling, the same legislation was not made to deal with the challenges associated with GM products. It provides for a similar legal standard for both conventional and GM products. The critics may argue that, once legislation provides for a legal standard to justify labeling for both GM and conventional products, then it would justify labeling of the former ones. But this argument is mistaken on a key point. The supporters of non-labeling do not defend the absolute non-labeling. What they, in fact, defend is that transgenics are submitted to the same labeling as the other products. The "non-labeling" represents this. As long as GM and conventional products are seen as "equivalent" in terms of risk, what they wish is that transgenic products are then labeled as conventional products. They defend that the consumer's legislation should be interpreted equally for both conventional and transgenic products once these products can be classified as "substantially equivalent". This implies the use of a single labeling standard for the products. What also implies the impossibility of differentiating the GMO products from the others.8
The moral argument presented by the alliance of precaution leads us to the idea of autonomy.9 Not only does this discourse not assume that consumers can guide their choices through risks, considering these choices as a right, but it also assumes that these choices can happen beyond reasons involving safety. For the groups who defend labeling in Brazil, safety was therefore just one of the important reasons but not the only one. In the civil lawsuit brought by IDEC, it informs that labeling has its right for factors which transcend a mere question of safety and then states that: "This data [gene of animal or vegetal species] is indispensable for the consumer perform his/her right of choice considering also allergenic, religious, cultural aspects" (IDEC apud PRUDENTE, 2000). This cultural view will be evident in the dispute over the scientific or the ideological character of precaution. As long as labeling has been associated with consumer's autonomy, its absence has been considered an attempt of his/her manipulation.
Between consumer's autonomy and manipulation: the ideological conflict about GMO labeling
Under the precaution discourse, the defense of consumer's choice and autonomy is associated with a view of his/her empowerment based on the consumer as an "economic person" who, by means of market tools, can now fulfill his/her choices. In this view, the consumer is sovereign and plays a predominant role in the way society's resources are allocated.10 The relevance given to labeling therefore is not only associated with the defense of consumer's choice but also with the influence that this choice can imply for the production and commerce of GM products. Labeling is seen as a means through which consumers can make their choices but can also, due to the same responsibility, reject these products. By doing so, consumers can subsequently influence the economic process more broadly. Non-labeling has been seen as a threat to consumer's autonomy as in its absence consumers can reveal confused beliefs about what they are consuming.11
Part of this confusion in consumer's information lies in the beliefs that they hold on governmental regulation. In the absence of labeling information of the products, consumers may belief they are not consuming GM products. The absence of labeling can make consumers conclude therefore that, by not having labeling, the products are not "GM products". This concern is expressed in the Brazilian case in the words of a representative when he says that the transgenic products in Argentina are being consumed here indirectly and without us knowing what we are consuming". This leads to "a disrespect to the consumer who should be at least informed that they are transgenics products" (GRANDÃO, 1999). Gabeira also points out that, once Brazil has been importing GM food, it would be "necessary that such food had an indication for consumers" because this process suggests that "Brazil continues to consume various types of genetically modified food without being aware of this" (GABEIRA, 2001). These remarks suggest that people should be consuming GM products not because they are in favor of them but simply because they are not aware they are consuming them.
Under the discourse of equivalence, the consumer is forbidden to choose between these types of products, at least while labeling for transgenics tend to be unqualified. After all, arguing against labeling or suggesting its irrelevance is to stand oneself against the possibility of the consumer's making this type of choice.12 So the discourse of equivalence involves, as we shall see next, a kind of political paternalism which exempts the consumer from the responsibility of decision making. According to this viewpoint, the process of rational choice is assigned to the entities and organizations which are considered more liable to take decisions on these matters. In the discourse of equivalence, the "consumer's right" was generally interpreted as right to information about food safety. What shows a consensus in this alliance is the assumption that, in terms of safety, there is no difference between transgenic and conventional soy. The acceptance of labeling became, in the beginning, a merely political issue to comply with the consumer's right. This position claims then that the communication provided by labeling should be a scientific communication strictly related to scientific facts. It also assumes that this scientific information is restricted to issues on food nutritional safety. These assumptions reflect the various lines which rule the American policy on labeling. So, we shall briefly examine some aspects of this policy and move to examine how these assumptions are reflected in the Brazilian case.
The policy of substantial equivalence: non-labeling as conventional labeling
The FDA policy in the USA claims that information on label is useful for the consumer only when it brings information on the food nutritional basis and on its implications to consumer's health. As pointed out by Pariza: "This position does not recognize a consumer's "right to know" simply for the sake "knowing", nor does it recognize a manufacturer's "right to inform" simply for the sake of "informing."(PARIZA, 2007:07). The FDA policy requires information when and only such information is important to issues involving product safety and consumer. But as long as, for the FDA, GM food is not even more or less safer than conventional food, this body does not require specific labeling for these products (STREIFFER and RUBEL, 2008).13 Under this perspective, it does not fall on labeling to express economic or cultural issues or any kind of information but strictly issues on food safety. If GM products do not endanger public health, transgenic labeling does not bring any useful information to the consumer. So labeling is seen as involving an arbitrary separation of "substantially equivalent" products.
The discourse of equivalence in Brazil reflects the lines of the American regulatory policy in many aspects. For example, when Bresser-Pereira declares that the government policy would follow the European policy, he also affirms that "in the case of soy, ( ) there is no substantial change of the product- the product is exactly the same ( ), the grain is exactly the same, undistinguishable" (BRESSER-PEREIRA, 1999). In this extract, the former minister was already aligning himself with the American policy. By saying that "there is no substantial change of the product" and that the "the product is exactly the same", Bresser-Pereira endorses the assumption of the substantial equivalence which is the basis of the non-labeling policy in the USA. The view of the former CTNBio, Esper Cavalheiro, on labeling also reflects the assumptions of the American standard, through which labeling should concentrate in the food nutritional components. Labeling, he says, "of any product should provide precise and correct information about the nutritional and compositional characteristics so as to ensure the consumer's free choice" (CAVALHEIRO, 2001). This shows that S&T and CTNBio started to defend labeling as from a precept that is exactly used for not labeling the GMO products in countries such as the USA.
Moreover, in the release period of RR soy, Bresser-Pereira (1999) also informed that "the American position was the same as Embrapa's". This statement can be regarded as a recognition that some governmental bodies were already aligned with the American standpoint for non-labeling GM products. And in the lack of any judicial decision in the period, it is possible to assume that they remained faithful to the precept of substantial equivalence. This position, in turn, contrasts with Bresser-Pereira's information when he says that "the Minister of Justice and I took the President of the Republic the position that we should have the European type labeling, which informs, whenever necessary, that the product contains a genetically modified product." (BRESSER-PEREIRA, 1999). The European policy for GM products could be, in this period, more different from the American policy in many aspects, but as far as labeling is concerned, it was "substantially equivalent". Whiteside (2006: 24) points out that "before 1997, EU regulations- like those in the United States - pertained only to the premarket testing of GMOs. It was assumed that once GMOs producers and various regulatory authorities concluded products were safe, then they were safe, period" [our emphasis]. The European position was, therefore, the same as the American position.
Defending the consumer from him/herself: political paternalism and the deficit of laypeople's knowledge
In the Brazilian case, non-labeling is justified as being based on consumer's "fears" and "suspicions". It is regarded as a way of correcting mistakes and misunderstandings that consumers themselves can make as to their consumption decisions. This is a typically paternalistic vision. It is assumed that non-labeling is based on the benefit of the consumer himself/herself. It would be a way to defend the consumer against his/her limitations in order to make the right choices in situations like these.
Paternalism is not something easily to be defined. Some definitions consider it as a restriction to an agent's freedom in favor of his/her own benefit. Other definitions emphasize the reasons through which this same intervention is made. Gerald Dworkin defines paternalism as the "interference with a person's liberty of action justified by reasons referring exclusively to the welfare, good, happiness, needs, interests, or values of the person being coerced" (DWORKIN apud GERT and CULVER, 1976: 45). So, paternalism can be understood as an interference of an agent's freedom of actions for the sake of his/her well-being.14 Following a legal interpretation of the conflict in the Brazilian case, this paternalism primarily expresses itself from a legal viewpoint. It emerges when it is suggested that CTNBio decisions should be followed assigning it almost absolute powers in its decisions.
"The law, by determining the regulatory limit, grants full compliance with the principle of precaution, followed in the biodiversity convention. The lack of scientific certainty cannot postpone the enforcement of norms, of rules. Automatically, by determining this process, this regulatory logic, creating a high level technical-scientific collegiate body to decide on whether the existence of risk or not, we are complying with the precepts of precaution." (José Silvino apud CESARINO, 2006: 103).15
And so, says the same technician, "if there is no risk, it [soy] will be treated as the common ones and goes to the vigilant bodies that originally have common authority for the common ones" (José Silvino apud CESARINO, 2006: 103). This argument summarizes the vision of the discourse of equivalence: for equivalent products, equivalent labeling. It is also worth noticing that, in this case, non-labeling (or conventional labeling) implies a situation in which it would be "complying with the precepts of precaution".
The idea that CTNBio is supported by law and that therefore its authority should be fully respected does not correspond to the existing differences between the knowledge based on CTNBio decision and the critics who rejected it. This paternalism expresses itself whenever the actors in favor of the release try to convince their interlocutors of their false beliefs about transgenic processes. In this case, the most important is not to know whether the public assigned all decisions to CTNBio or not but rather whether is this approval would be based on the public's own knowledge. Or else, as pointed out by Streiffer and Rubel (2008) in the American case, paternalism implies that the public would assign FDA the decision if "it was well-informed" as it is assumed that if "were people better informed, they would change their preference, thereby giving hypothetical consent to the delegation" (STREIFFER AND RUBEL, 2008:31).
Take the case of CTNBio president for an example of this case. When asked about the relation of trust between the public and the regulatory agencies, Walter Colli, on a debate held by the Research Support Foundation of State of São Paulo (in Portuguese, Fapesp) on 10 May 2008, answered the question involving labeling and the relation of trust between scientists and laypeople as follows:
"When you eat organics, you are eating Bacillus and you are eating this gene, the same thing! Then the only thing I want is that you understand what a scientist is saying, that's all. If [you are] for or against, it is the same thing as being corinthiano or palmeirense, I am corinthiano. What am I going to do?" (COLLI, 2008: 34)
CTNBio president's answer was not restricted to this, but this was the point of his argument: transgenic and organic products are even similar in certain circumstances. It is surprising then how government representatives, CTNBio, industries and the political sector try to demonstrate the equivalence of both GM and conventional products since this equivalence is kept away from the principle that rules the transgenic labeling policies in various countries, which assume the existence of the non-equivalence of these products. The public's suspicious concerning the scientists is seen as the result of some information deficit and the central interest then is to make the other understand "what the scientist is saying". In this case, the aim is to make the public understand that transgenics are not so much different from conventional products and that, for this same reason, there is no reason for concern. It is assumed that if people were better informed, they would agree with CTNBio decisions.
GMO Traceability and segregation: elements of an environmental utopia?
To conclude this analysis, it is important to discuss one last issue: the way the traceability and segregation systems have been incorporated to the debate. Smith and Phillips (2002) make a distinction between systems of identity of preservation, segregation and traceability that help us to understand the Brazilian conflict in some points. As claimed by the authors, segregation can be seen as a "a regulatory tool that is required for variety approval and commercial release of grain and oilseed varieties that could enter the supply chain and create the potential for serious health hazards" (SMITH and PHILLIPS, 2002: 31). Segregation occurs where, for food safety measures, there is a concern about the mixture of the segregated product in relation to all other products. This segregation can be summarized as follows: "systems of IPPM are used to capture premium, and segregation is used to ensure food safety" (SMITH and PHILLIPS, 2002:31). The system of traceability leads us to the same question. Smith and Phillips (2002) argue that traceability systems are used when "unacceptable bacteria levels or intolerable levels of pesticide or chemical residues need to be quickly and completely removed from store shelves. Traceability systems allow for retailers and the supply chain to identify the source of contamination and thereby initiate procedures to remedy the situation" (SMITH and PHILLIPS, 2002:31). Thus, EU sees traceability as providing for "a "safety network" that would allow vigilance on unforeseen adverse effects" (EU apud SMITH and PHILLIPS, 2002: 32).
This makes us understand why concerns about the systems of segregation and traceability have been away from government sectors, CTNBio and the industry. These systems are nothing less than the result of a policy of food safety for certain products that present a not well known risk level. Therefore, if transgenic soy is seen as presenting a safety level equivalent to the conventional soy- what the alliance of equivalence has been defending during all the conflict- what would be the reason for the implementation of such kind of systems in Brazil? Insisting in these systems would be incoherent when it is said that the GM product is completely safe and when it is claimed that labeling is a simple political issue and not a safety one. It will not be then a coincidence to examine that, in over 10 years of conflict, the government, CTNBio and the industry sectors have not offered any detailed program for the segregation and traceability of GM products. The creation of systems of food safety with these characteristics has been supported by environmentalists and particularly organizations such as IDEC and Greenpeace.
Final remarks
The Brazilian conflict about labeling evidences how this issue can be interpreted distinctively depending on which side one is. For supporters, it has been associated with environmental safety and values which transcend the simple consumer's right. It has been also associated, as seen, with the principle of precaution and the creation of a system of food safety involving segregation and traceability. This stance has been confronted with the one offered by the government, CTNBio and the industry. Not only did the arguments presented by these actors give some doubtful support to "consumer's right" suggesting many times that this could be complied with conventional labeling, but they were also based on the principle of substantial equivalence which, in other countries, is used precisely to invalidate the requirement of specific labeling for GMOs. The principle of "substantial equivalence" is on the basis of many of the arguments that reject GMO labeling. However, the labeling of GM products assumes a gap with this principle once these policies are based on the assumption that such products are not substantially equivalent. Among the countries with specific labeling legislation, the only aspect in common is the almost generalized conviction that GM products, as Greure e Rao (2007:52) point out, "are no substantially equivalent to their conventional counterparts". In these countries, labeling is seen as compulsory because it is known that "consumers should be informed of the novel traits and properties of the food products in order to make informed decisions (GREURE and RAO, 2007:52). The application of the principle of substantial equivalence is therefore ineffective to support a transgenic labeling policy. The principle invalidates a basic assumption of the GMO labeling policy which informs that these products are different from the others and that, for this same reason, also deserve a different treatment. It is this difference that provided for the creation of the Biosafety Law in the country and the own creation of CTMBio which, ironically, turned out to treat RR soy as an equivalent product as the others.
References
ANBIO. Report on the commercial approval of transgenic soy in Brazil. Available at <http://www.anbio.org.br/notícias/relatório.htm> on 23/7/2008.
BICK, Léo F. Public Hearing n. 2036/05, held by the Commission of Environment and Sustainable Development on non-labeling products sales in Brazilian supermarkets. Held on 08/12/2005.
BRESSER-PEREIRA, Luiz C. Alimentos transgênicos e biossegurança. Available at <http://www.bresserpereira.org.br/view.asp?cod=596> on 02/03/2008.
BRESSER-PEREIRA, Luiz C. Public Hearing 053/99, held by the Commission of Consumer Rights, Environment and Minorities. Debate on the authorization of transgenic food for production and consuming in the country. autorização para produção e consumo de alimentos transgênicos no país. Held on 16/6/99.
CASTRO, Luiz A. B. de. Public Hearing 0927/00, held by the Commission of Consumer Rights, Environment and Minorities. Held on 13/9/2000.
CASTRO, Luiz A. B. de. Public Hearing n. 0927/00, held by the Commission of Consumer Rights, Environment and Minorities. Discussion on the labeling of transgenic products. Held on 13/9/2000.
CAVALHEIRO, Casper. Transgênicos: sociedade precisa de informação para decidir. Jornal da ANBio, 1 (4), 2001, pp. 01.
CESARINO, L.M.C. N. Acendendo as luzes da ciência para iluminar as luzes do progresso. Thesis (Master's Degree). Programa de Pós-Graduação em Antropologia Social. Universidade de Brasília, Brasília, 2006.
CEZAR, F. G. Previsões sobre tecnologias: pressupostos epistemológicos na análise de risco da soja transgênica. Dissertação (Mestrado) - Departamento de Filosofia, Universidade de Brasília, Brasília, 2003.
CHRISTOFOROU, Theofanis. Genetically modified organisms in European Union law. In: SADELEER, Nicolas de (ed.) Implementing the precautionary principle. Approaches from the Nordic Countries, EU and USA. UK: Earthscan, 2007, pp. 197-228.
COLLI, Walter. Transgênicos e mídia. Transcription of the debate between Walter Colli and Herton Escobar. Held on 10/5/2008. Available at <http://www.revista. pesquisa.fapesp.br/pdf/revolução_genomica/ colli.pdf>on 04/4/2008.
DEGNAN, Fred H. Biotechnology and the food label. In: WEIRICH, Paul. Labeling genetically modified food. New York: Oxford University Press, 2007, pp. 17-31.
FISCHER, Frank. Reframing public policy: discursive politics and deliberative practices. Nova York: Oxford University Press, 2003.
GABEIRA, Fernando. Public Hearing n. 000565/01. Special Commission - PL No. 2905/97 - Genetically modified food. Held on 19/6/2001, pp. 1-24.
GABEIRA, Fernando. Public Hearing n. 0533/99, held by the Commission of Consumer Rights, Environment and Minorities. Debate on the authorization of transgenic food for production and consuming in the country. Hold on 16/6/1999.
GERT, Bernard e CULVER, Charles M. Paternalistic behavior. Philosophy and Public Affairs, 6 (1), 1976, pp.45-57.
GREURE, G. P. and RAO, S. R. A review of international labelling policies of genetically modified food to evaluate India's Proposed Rule. AgBioForum, 10 (1), 2007, pp. 45-57.
GUIVANT, Julia S. A governança dos riscos e os desafios para a redefinição da arena pública no Brasil. In: Centro de Gestão e Estudos Estratégicos. Ciência, Tecnologia e Sociedade. Brasília: CGEE, 2005, pp. 47-85.
GUTHMAN, Julie. Eating risk. The politics of labeling genetically engineered foods. In: SCHURMAN, Rachel et al. Engineering trouble. Biotechnology and its discontents. Berkeley: University of California Press, 2003, 130-151.
HAJER, Maarten. The politics of environmental discourse: ecological modernization and policy process. Oxford: Clarendon Press, 1995.
IDEC. Alimentos transgênicos: a posição do IDEC. Available at <http://www.aao.org.br/jornal07.pdf.> on 15/3/2008a.
IDEC. Transgênicos. Available at <http://www.IDEC.org.br/files/relatório_transgênicos.doc.> on 13/11/2008b.
IDEC. IDEC contesta declarações da Abia. Available at <http://www.IDEC.org.br/emacao.asp?id=854.> on 07.11.2008c.
JACOBS, Michael. Green economy. London: Pluto Press, 1991.
KLINTMAN, Mikael. The genetically modified (GMO) food labeling controversy: ideological and epistemic crossovers. Social studies of science, 32 (1), 2002, pp.71-91.
MOURA, Confúcio. Public Hearing n. 000565/01, held by th Commission of Consumer, Rights, Environment and Minorities. Discussion on the projec of law no. 2905 under review by the Special Commission. Held on 19/06/2001, 01-24.
ODA, Leila M. Presidenta da CTNBio fala sobre a rotulagem dos transgênicos. on 12/11/08.
ODA, Leila M. Para presidente da CTNBio, Leila Oda, é inevitável a entrada de transgênicos e a rotulagem dos produtos é a garantia do consumidor. Story published on the newspaper Valor Econômico, 5 June 2000. Available at <http://www.agrisustentável.com/trans/dialoda.htm.> on 10/4/2008.
PARIZA, Michael W. A scientific perspective on labeling genetically modified food. In: WEIRICH, Paul. Labeling genetically modified food. New York: Oxford University Press, 2007, pp. 03-09.
PIZZATO, Luciano. Public Hearing n. 0927/00, held by the Commission of Consumer Rights, Environment and Minorities. Discussion on the labeling of transgenic products. Held on 13/9/2000, pp. 01-59.
PRUDENTE, Antonio S. Transgênicos: decision on public civil lawsuit. Decision on 26/6/2000. Available at <http://jus2.uol.com.br/pecas/texto.asp?id=337>on 11/2/2009, pp. 1-62.
RUBEL, A. and STREIFFER, R. Respecting the autonomy of European and American consumer: defending positive labels on GMO Foods. Journal of Agricultural and Environmental Ethics, 18 (1), 2005, pp. 75-84.
SILVER, Lynn. Public Hearing n. 001019/01, held by the Commission of Consumer's Rights, Environment and Minorities. Held on 27/9/2001.
SILVER, Lynn. Debate. In: SOARES, Francisco de A. P. (org.). Anais do seminário. A sociedade frente à biotecnologia e os produtos transgênicos. Brasília: Confea, 1999, pp. 47-53.
SMITH, Stuart and PHILLIPS, Peter W. B. Product differentiation alternatives: identity preservation, segregation and traceability. AgBioForum, 5(2), 2002, pp: 30-42.
STREIFFER, Robert e RUBEL, Alan. Democratic principles and mandatory labeling of genetically engineered food. Available at <http://www.tccouncil.org/reports/Streifferand Rubel%20GELabeling.pdf.> on 10/4/2008.
STREIFFER, Robert and RUBEL, Alan. Genetically engineered animals and the ethics of food labeling. In: WEIRICH, Paul. Labeling genetically modified food. Nova York: Oxford University Press, 2007, pp. 63-87.
WHITESIDE, Kerry H. Precautionary politics. Principle and practice in confronting environmental risk. Cambridge: The MIT Press, 2006.
1 Cristiano Luis Lenzi has a Ph.D. in Social Sciences- UNICAMP. Professor at the School of Arts, Sciences and Humanities (EACH-USP). E-mail: clenzi@usp.br.
2 The fact that the government, CTNBio and the industry stand, in the conflict, in favor of labeling does not imply that their representatives were effectively for compulsory and specific labeling for transgenics. As we will try to demonstrate, the signal given when one is for transgenic labeling can simply suggest that one is being in favor of conventional labeling for these products. In this case, although transgenics can be labeled, such option does not provide for the differentiating GM products from conventional ones.
3 Lynn Silver, IDEC representative, refers to the following dialogue with CTNBio president: "In fact, what I understood from Dr. Barreto de Castro's speech, it was not clear if this was his personal position or the commission's position but that, not being identified any risks for human health, it was irrelevant the inclusion of labeling in the indicative of genetic engineering" [our emphasis] (SILVER, 1999: 53).
4 Similarly to the representative below, the former S&T minister Bresser-Pereira will state that arguing simultaneously that the government would follow the European labeling policy in which "you show, whenever necessary, that the product contain genetically modified product" (BRESSER-PEREIRA, 1999). However, as we shall see next, this statement encloses a strong contradiction when examined in the context into which the European labeling policy for GMOs has been developed.
5 Thus, for representative Luciano Pizzato: "We have genetically modified trees. Why do we only discuss agricultural products? Why has the Department (
) not required that the genetically modified medicine warn in the labeling that they are transgenics? Why does this Commission forget that?" (PIZZATO, 2000: 16).
6 The argument shows once again as CTNBio position continued to have strong influence to invalidate labeling. This last one was unnecessary due to the fact that transgenics products have been "evaluated and considered safe by the National Technical Commission of Biosafety" (CTNBio). The argument of former CTNBio president, Leila Macedo Oda, that labeling is a "much more a political question than a technical one" hides then the fact that the scientific considerations used by CNTBio to release soy were repeatedly used to try to discredit the need of labeling of GM products in the country. The political debate on labeling has been in any moment dissociated from the scientific controversies that involve its commercial release.
7 On a report presented by the National Biosafety Association (in Portuguese, ANBio) it is informed that "judge Selene's account makes it clear that there are no reasons of technical-scientific or legal nature which prevent the commerce of RR soy in Brazil approved by CTNBio 54 notice" [our emphasis] (ANBio, 2008).
8 Let us consider, for example, the following information of former CTNBio president, Leila Oda. According to her: "We will not label to say the product is dangerous but to respect the consumer's right of option wherever its reasons. If a person is allergic to egg, he/se has the right to know that the final product, transgenic or not, contains egg protein" [our emphasis]. It is worth informing that a product containing "egg protein" does not imply to inform that the product is transgenic or not. As far as the nutritional composition is seen here as the main goal of labeling, what would be informed to the consumer, in this case, is no the way it was produced (GM product) but only its nutritional components.
9 Autonomy is related to a person's capacity to make his/her own decisions from the values which comprises his/her way of living. Autonomy does not refer only to the capacity of "making choices" but to the capacity of making choices which harmonize with the values that guide a certain way of living (RUBEL and STREIFFER, 2005).
10 For the case we are examining here, this vision of "economic person" only suggests the consumer's capacity to use his/her power as a means of influencing investments and productive processes. This was the meaning given by the economist Ludwig Edler Misses when he said that "the consumers are the masters, to whose whims the entrepreneurs and capitalists must adjust their investments and methods of production" (MISSES apud KLINTMAN, 2002:73).
11 In 2003 in the USA, although 70% of processed food had GM ingredients, 58% of consumers believed that they had never consumed transgenics. In 2004, 41% of consumers were not sure whether the genetically modified food was available in supermarkets while 11% of them believed that they were not. Still in this period, 46% of consumers were not sure whether they had consumed GMO products and 23% believed that they did not. In another research conducted in the USA, only 33% of consumers knew that labeling was not required for GMO products and 28% believed erroneously in the compulsory labeling requirement for GMO products. More about this issue, see Streiffer and Rubel (2008).
12 As Jacobs (1991: 43) says, "consumers have to be offered a choice to express their environmental concerns. (...) Although orthodox economists may pretend that consumers have "sovereignty" in a market, one can actually only buy what is offered by producers. Although orthodox economists claim that consumers have "sovereignty" in the market, in fact one cannot buy more than what producers offer. On the other hand, as pointed out by Klintmann (2002), this marginal role of consumer is not always consistent with the idea of free market. This can happen once this passivity can be accepted as the argument that consumer's sovereignty is only valid under circumstances where consumers "rule" using valid information. In the Brazilian case, the arguments against labeling due to consumers' "fears" follow this viewpoint.
13 In 2000, asked about the reason of not labeling GMO products, FDA in the USA made the following statement:: "We are not aware of any information that foods developed through genetic engineering differ as a class in quality, safety, or any other atribute from foods developed through conventional means. That's why there has been no requirement to add a special label saying that they are bioengineered. Companies are free to include in the labeling of a bioengineered product any statement as long as the labeling is truthful and not misleading. Obviously, a label that implies that a food is better than another because it was, or was not, bioengineered, would be misleading". (apud DEGNAN, 2007: 27). The FDA policy reflects a change in labeling policy happened in the industrialized countries in the last decades. The FDA policy is based on a sound science approach which reduces information to be placed in labels to scientific information related to issues of nutritional safety. From a space reserved to product advertisement, labeling has changed in the USA into a means of providing consumers with scientific information. In this process, the roles of the regulatory agencies have changed. More than ensuring the product contents, the agencies started to monitor the truth of the information contained in labeling. Cultural issues or issues involving the process through which the products are made are excluded from this labeling system (GUTHMAN, 2007).
14 In the American case, one way of defending oneself non-labeling is based, for example, on paternalistic grounds. This paternalism can be expressed as follows: the public assigns FDA the decisions on labeling because: (1) the public elected the Congress; (2) which created FDA, a legitimate authority; (3) conventional labeling is embedded in legitimate labeling. Therefore, the public would agree with the current FDA policy. For discussion on paternalism in the American case, see Streiffer e Rubel (2008).
15 This paternalism is also supported in Bresser-Pereira's vision when He says that "the National Congress has approved the Biosafety Law and this law created the National Biosafety Technical Commission (CTNBio). (
). It is CTNBio responsibility then to examine, case by case, whether a certain product is liable or not of approval for health and the environment on the viewpoint of biosafety. (
) The most recent example of the Commission authority to implement the Biosafety Law was the approval and enforcement of the commercial use of transgenic "round up ready" soy. This product was deeply analized and was finally approved by CTNBio. Therefore, the policy of the National Congress concerning transgenic products is being rigorously complied with" (BRESSER-PEREIRA, 1999).